Search
A Philosophical Inquiry into the Nature and Problems of Religious Experience
DOI: 10.17160/josha.7.2.663
The paper aims at studying the nature and problems of religious experience. Religious experience is defined as a subjective experience which is interpreted in a religious circle as an encounter with God or gods or higher-order realities. The researcher identifies the features of religious experience and various activities that can enhance its manifestations. The philosophical problems of religious experience are also discussed. Despite the challenges of religious experience, the writer concludes that the experiences cannot be totally disregarded, but have to be examined discreetly in order not to be deceived, as faith without reasoning leads one to errors, deceptions and wrong belief. The work is qualitative research and the method used is philosophical analysis.
Demetrios Award 2020
DOI: 10.17160/josha.7.2.662
The JOSHA Team is very pleased to share information with you about our Demetrios Prize 2020. The International Academy of Sciences, Humanities, and Arts (IASHA e.V.), with the support of the BioThera-Roland Mertelsmann Foundation, will award three prizes of 500 euros for the categories BACHELOR, MASTER and DOCTOR THESIS. Our editors will make the selection and the theses of the winners will be published in JOSHA and can be referenced with their DOI number! The “Journal of Science, Humanities, and Arts – JOSHA” has been initiated to create a novel internet platform to access the broad diversity of important discoveries and creativity in the fields of Science, Humanities, and Arts. At JOSHA we believe, that "Knowledge that is not communicated is wasted knowledge".
Immortal
DOI: 10.17160/josha.7.2.656
This is one of the Articles "El Gato y La Caja" (Buenos Aires, Argentina) shares with the JOSHA Journal. This time the English version of the original article. In the field of Life Sciences, the author presents an incredible story about Henrietta Lacks, a woman that changes and improves forever all the labs around the world. Shortly after being diagnosed (and without her consent) a small piece of Henrietta Lacks´ tumor was cut out and put it in a glass dish. Surprisingly, her uterine-cervical cancer cells had the ability to grow indefinitely. In this single immoral act, the beginning of a revolution in cell biology had occurred. On October 4, 1951, Henrietta Lacks died without knowing that a little piece of her was going to be found in almost every laboratory in the world that uses cell culture. This is her story.
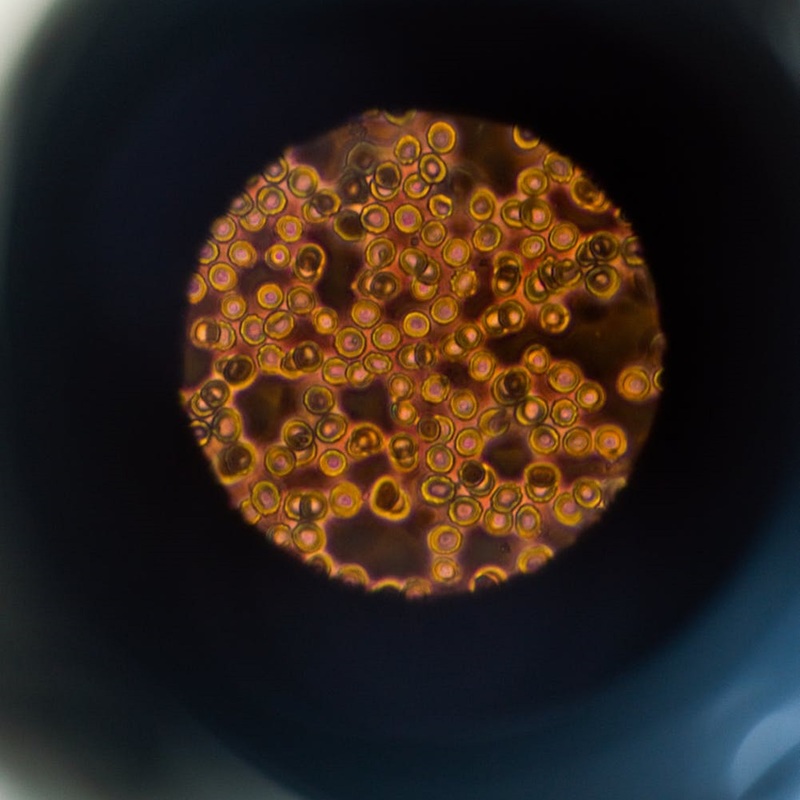
Automatic Breast Cancer Cell Classification using deep Convolutional neural Networks
DOI: 10.17160/josha.7.2.652
Automated cell classification in cancer biology is an active and challenging task for computer vision and machine learning algorithms. In this Thesis, we first compiled a vast data set composed of JIMT-1 human breast cancer cell line images, with and without therapeutic drug treatment. We then train a Convolutional Neural Network architecture to perform classification using per-cell labels obtained from fluorescence microscopy images associated with each brightfield image. The study revealed that our classification model achieves 65% accuracy in breast cancer cells under chemotherapeutic drug treatment with doxorubicin and paclitaxel. Furthermore, it reached 70% accuracy when classifying breast cancer cells without drug treatment. Our results highlight the potential of machine learning and image analysis algorithms to build new diagnosis tools.
Research on genetic Databases: Duties of Rescue and equal Respect to all
DOI: 10.17160/josha.7.2.650
The ideal exercise of Castellanos et al. asks IRB members to accept or deny a researchers’ request for disclosing genes associated with breast-ovarian cancer, a potential clinically significant finding (CSF), to a subgroup of 1500 research participants of a research database of 300,000 subjects. Here, we defend that researchers are under a rational duty to rescue. However, we also argue that only a decision that equally respects the rights and interests of all participants could be a fair and reasonable decision, compatible with ethics and the rule of law. Consequently, IRB members should not only accept the researchers’ request to disclose potential CFS to database participants with breast-ovarian cancer genes but also to advocate for an update in the disclosure policy for all participants of the genetic research database.
Systemic Relevance - of Culture and Freelancers in Times of unpredictable global Events: The Corona Crisis
DOI: 10.17160/josha.7.2.649
COVID-19 has brought the world to its knees, but it has brought freelance workers and artists to bankruptcy. Creativity is needed now more than ever to solve the problems of the day. Franziska Buttgereit is a mezzo-soprano who has been a member of the Freiburg Theatre's extra choir since 2010 and its first director since 2017. In 2015 she began her singing studies with Prof. Christiane Libor at the Musikhochschule Schloss Gottesaue in Karlsruhe and has been collaborating with the University of Music and the Stadttheater Freiburg, which has led to a small role in performances and the DVD production of the same name "Cendrillon" by Jules Massenet.
Thinking with Others. A survival Guide in times of post-Truth - Of Words and Things, Chapter 1
DOI: 10.17160/josha.7.2.646
Below is the introduction, preface and first chapter of the book Pensar con otros. Una guía de supervivencia en tiempos de posverdad (Thinking with others. A survival guide in times of post-truth), published in 2018. A book that aims to be a starting point to break down post-truth into its main components through concrete examples, as a way to better prepare ourselves to identify the structures that foster post-truth and thus achieve, together, survival and victory. Post-truth allows facts to become facts as long as they fit the desires of each group, of each tribe. Each of these groups develops its own narrative with a language that privileges the capacity to provoke emotions, and pushes those emotions to build landscapes only accessible to those who share the way of looking at them. This discontinuity in the landscape is a threat to the existence and development of meaningful human bonds, to our coexistence as a species on this planet and, thus, to our survival.
Stage fright - blessing or curse?
DOI: 10.17160/josha.7.2.645
Whether it be a surgeon or a performing artist, most people experience stage fright at some point in their lives. Varying levels of anxiety put stress on the body and impact one’s ability to perform, however, the ability to cope with a given situation leads directly to positive or negative outcomes. Franziska Buttgereit is a mezzo-soprano who has been a member of the Freiburg Theatre's extra choir since 2010 and its first director since 2017. In 2015 she began her singing studies with Prof. Christiane Libor at the Musikhochschule Schloss Gottesaue in Karlsruhe and has been collaborating with the University of Music and the Stadttheater Freiburg, which has led to a small role in performances and the DVD production of the same name "Cendrillon" by Jules Massenet.
Architectural Design Process: Consulting with Nature
DOI: 10.17160/josha.7.2.643
In the continuum of evolution and adaptation of living organisms, ranging from microorganisms to human beings, there are certain cycles of time that carry with its cardinal changes that human beings must face and find the best possible harmony of eligibility. The new millennium brings with it a new lifestyle as a result of a number of determining factors, driven by technology development and the phenomenon of globalization. Therefore, we have come into a situation where a number of questions need to be raised about the necessity of redefining many daily life habits. The study presented in this paper investigated the architectural design process to consult with nature, where to many urban structures are being ruined in the name of sustainable architecture. We are constantly experiencing original situations known by intuition, but we have forgotten them, remembering the same thing can be ten times faster.
Editorial Volume 7, Issue 1
DOI: 10.17160/josha.7.1.641
Dear JOSHA readers, once again we find ourselves saying goodbye and closing another issue. This time it is the first one for the year 2020, which is full of important changes for our non-profit journal. Among other things, we must inform you all that our editorials will be published every two months, that is, from 2020 all our issues will be published bimonthly, always on the 30th of every second month. We also like to remind that you can upload a video link of YouTube to your article!








.jpg?1583079463)